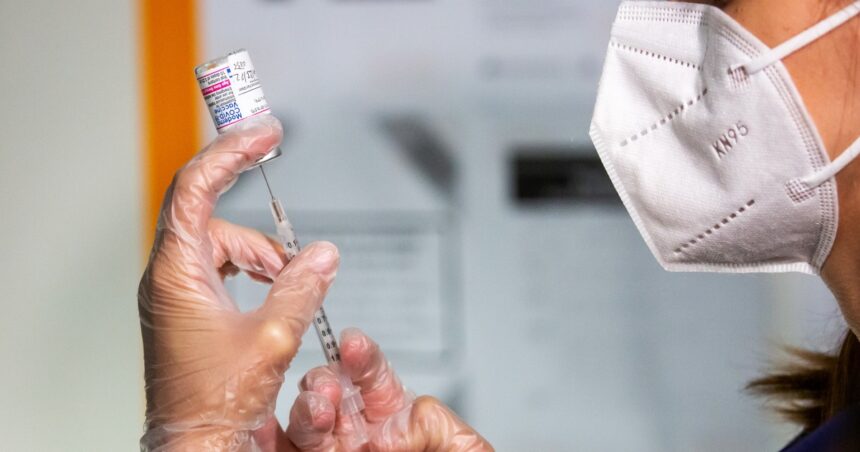
Food and Drug Administration (FDA) officials are set to present claims linking the Covid vaccine to 25 deaths in children at an upcoming meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP). The meeting is anticipated to generate significant public interest and scrutiny. Reports indicate that the FDA’s allegations are based on data derived from the Vaccine Adverse Event Reporting System (VAERS), which is a database managed by the FDA and the CDC.
However, concerns have arisen regarding the interpretation and utilization of VAERS data. The database allows anyone—including medical professionals, patients, and caregivers—to report adverse events they suspect are related to vaccinations. Critics argue that these reports are often unverified and highlight the need for caution in drawing definitive conclusions from such data. Dorit Reiss, a vaccine policy expert at the University of California Law, San Francisco, emphasized that VAERS reports alone cannot establish a direct causation between vaccination and deaths, asserting that comprehensive studies comparing affected individuals with those unvaccinated are essential for such determinations.
Andrew Nixon, a spokesperson for the Department of Health and Human Services, mentioned that the FDA and CDC routinely analyze VAERS along with other safety data, with findings shared through the established ACIP process. Meanwhile, FDA Commissioner Marty Makary noted the ongoing investigations into reported deaths among children following vaccination, stating that detailed findings would be disclosed soon.
The VAERS platform itself cautions that the case reports can contain inaccuracies or bias, underlining that scientific use of the data necessitates careful interpretation. The Washington Post reported that Dr. Tracy Beth Høeg, a special adviser to Makary and a vocal critic of Covid vaccinations for children, will present the findings at the upcoming ACIP meeting.
Former FDA officials have also voiced skepticism about the claims being made. One anonymous official asserted that all autopsy reports were thoroughly reviewed and did not support the alleged links to the vaccine. In contrast to the emerging claims, multiple studies have shown that Covid vaccines are safe for children and significantly reduce the likelihood of hospitalization and death. A 2023 analysis published in JAMA Pediatrics encompassing over 10 million vaccinated children confirms the vaccines’ efficacy and safety profile compared to unvaccinated peers.
Further studies have indicated no increased risk of adverse events in younger populations receiving the Covid shots, aside from a minor elevated risk of myocarditis in adolescent males following specific doses. During previous FDA advisory meetings, Pfizer provided real-world data showcasing the safety of its vaccine, noting ongoing post-approval studies involving over 60 million individuals globally.
The debate around vaccine safety has been fueled by anti-vaccine activists who frequently point to VAERS data to argue that vaccines pose a significant danger. Health and Human Services Secretary Robert F. Kennedy Jr., a prominent anti-vaccine advocate, recently highlighted the number of reports linked to the Covid vaccine during a Senate Finance Committee hearing, claiming that they surpass the tally for all vaccines in history—including during extensive historical data collection.
In a controversial move, Kennedy replaced all members of the ACIP with individuals selected by him, some known for anti-vaccine leanings. This restructuring has prompted criticism from the American Academy of Pediatrics, which condemned the new appointments as a significant deviation from the committee’s focus on children’s health. Among the appointments, Retsef Levi, a non-medical professor from the Massachusetts Institute of Technology, has voiced concerns about potential harms from Covid vaccines, particularly for children.
Kennedy has also limited access to the newly approved fall Covid vaccines, targeting primarily those aged 65 and older or individuals with underlying health conditions, thereby causing confusion among patients and pharmacies. In recent commentary, Makary aligned the FDA’s approach with those of other nations, advocating the necessity of assessing whether the benefits of successive vaccine doses outweigh inherent risks, particularly for healthy children who may have already contracted the virus. The discussions and recommendations arising from the ACIP meeting next week are poised to play a crucial role in shaping future vaccination protocols and public health messaging surrounding Covid-19 vaccination for children.